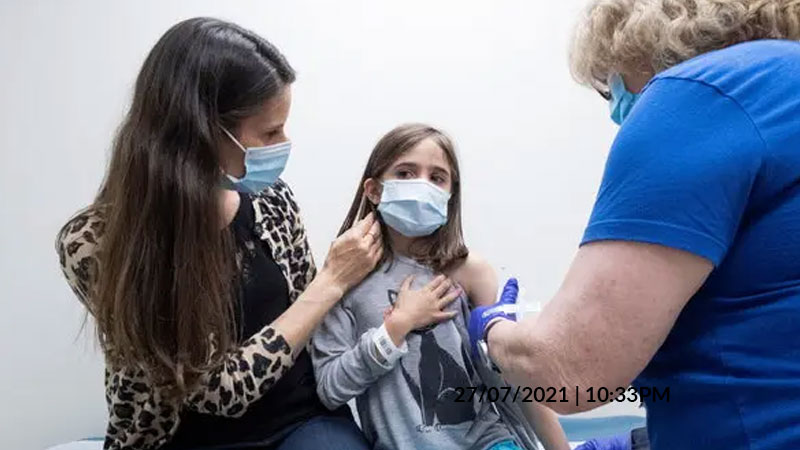
At the urging of federal regulators in the US, two coronavirus vaccine makers are expanding the size of their clinical trials for children aged 5 to 11 years — a precautionary measure designed to detect rare side effects including heart inflammation problems that turned up in vaccinated people younger than 30 years.
US President Biden promised at a meeting in Ohio last week that emergency clearance for pediatric vaccines would come “soon,” but the White House has not been specific on the timeline.
The New York Times says it was unclear whether expanding the studies will affect when vaccines could be authorized for children.
The Food and Drug Administration has indicated to Pfizer-BioNTech and Moderna that the size and scope of their pediatric studies, as initially envisioned, were inadequate to detect rare side effects.
Questions about vaccinating children — including those under 12 — are of huge interest to parents and teachers. Regulators will be required to balance potential side effects of coronavirus vaccination against the risks of COVID-19.
Members of a C.D.C. advisory committee have said that the benefits of shots for people older than 12 greatly outweigh the risks, including of heart problems.
The F.D.A. has asked the companies to include 3,000 children in the 5-to-11-year-old group, the group for whom results were expected first, according to people familiar with the situation.
One of the people, granted anonymity to speak freely, described that figure as double the original number of study participants.
A spokesman for Moderna, Ray Jordan, confirmed that the company intends to expand its trial “to enroll a larger safety database which increases the likelihood of detecting rarer events” and expects to seek emergency authorization late this year or early next year.
The Moderna trial began recruiting patients in March with the aim of enrolling 6,795 participants younger than 12.
The participants were to be split equally into three age brackets, including a 6 to 11 year old group, of 2,265 participants each.
Jordan said the company is “actively discussing” a proposal with the F.D.A. to expand the trial.
Pfizer is on a faster timetable than Moderna, and may be able to meet the F.D.A.’s expectations on a bigger trial size and still file a request to expand emergency authorization of its vaccine by the end of September. Reviewing all the safety and efficacy data will likely take regulators at least a few weeks.
Pfizer has previously said it expects to have results for the 5-to-11-year-old group in September, with results for children aged 2 to 5 shortly after that. Results for the youngest children — 6 months to 2 years old — are expected in October or November. A spokeswoman said that the company had no updates on its timetable.
Many public health experts argue that, with so much attention focused on hospitalizations and deaths among older Americans infected with the coronavirus, the risk for children has been overlooked.
More than four million American children and adolescents have tested positive for the virus since the onset of the pandemic, the American Academy of Pediatrics reported last week. Of those, at least 346 have died.
[Source: The New York Times]
Stay tuned for the latest news on our radio stations